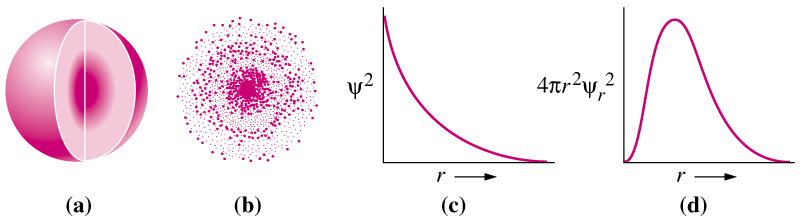
The eigenfunctions of the Schrödinger equation form a basis for the state space of the system. A more general formulation Quantum state vectors No other sources of energy exist in the system as defined, and so the particle's state must be some linear combination of the eigenstates of H. The first term of the Hamiltonian corresponds to the classical expression for kinetic energy, p 2/2 m (see above), and the second term is the potential energy as defined. The Hamiltonian essentially contains all of the energy "sources" of the system, and its eigenstates describe the possible state of the system entirely.Īs an example, consider a particle in a one-dimensional box with infinite potential walls and a finite potential a inside the box. In other words, the state of any system is reducible to a linear combination of solutions of the Schrödinger equation for that system. Also, the various eigenfunctions ψ n must be linearly independent and in fact form a basis for the state space of the system. It must be a self-adjoint operator because its eigenvalues E are the discrete, real energy levels of the system. H is an example of a quantum-mechanical operator, the Hamiltonian (classical Hamiltonians also exist). Indeed, it is generally assumed that the (time-dependent) Schrödinger equation itself is one of the central axioms of quantum mechanics. Note that the heuristic reasoning in this section makes the form of the Schrödinger equation plausible, but should not be seen as a formal derivation departing from certain axioms. In spherical coordinates, the spherical definition of the Laplacian gives In three dimensions, the second derivative becomes the Laplacian:
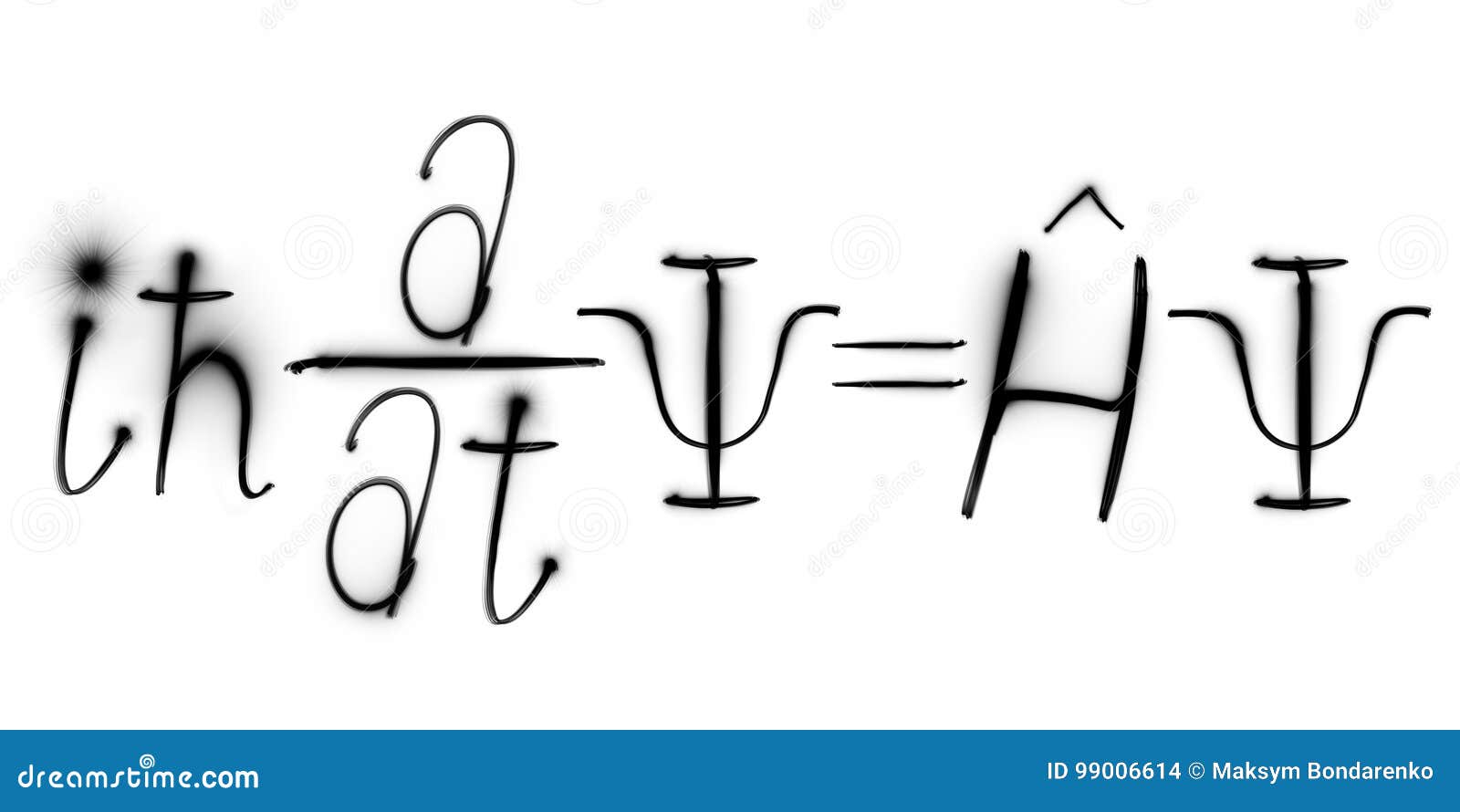
When the probability amplitude of the wavefunction is independent of time, it can be shown that energy is constant, and so the equation reduces to

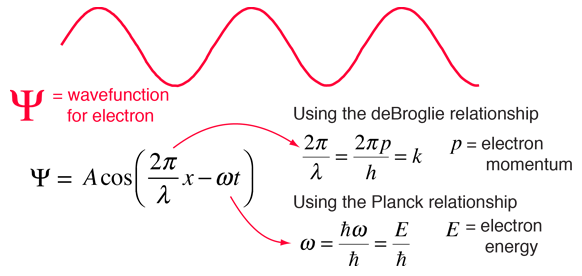
Substituting for p and E yields the one-dimensional time-dependent Schrödinger wave equation, Using the classical relationship between kinetic energy and momentum and allowing for a potential, Where is Planck's constant divdided by 2π.īased on the functional form of ψ, we see that The absolute square |ψ| 2 of this function (its probability amplitude) is a constant independent of position and time, which makes sense for a constant beam of particles: there is an equal probability of finding a particle at every point along the beam and any time. We start by assuming that a beam of particles will have a wavefunction of the form But what is the form of Schrödinger's equation, which describes the time and position evolution of the wavefunction? The wavefunction describes a wave of probability, the square of whose amplitude is equal to the probability of finding a particle at position x and time t. In the case that the quantum system has a classical counterpart, the Hamiltonian operator is obtained by replacing the terms in the classical Hamiltonian function by linear operators. Both Schrödinger equations contain an energy operator H, called a Hamiltonian. In many cases these energies can have only certain discrete values, which one expresses by stating that the energies are quantized.īuilt into the eigenvectors are the probabilities of measuring all values of all physical observables, meaning that a solution of the Schrödinger equation provides a complete physical description of a system. Mathematically, the time-independent Schrödinger equation is an example of an eigenvalue problem whose eigenvectors ψ are called "wavefunctions" or "quantum states" and whose eigenvalues E correspond to energy levels. It should also be noted that the fact that the time independent equation takes the form of a simple eigenvalue problem (thus being more amenable to mathematical analysis) that makes it so useful. The conditions that must be met to enable this separation of variables arise very frequently in chemistry and physics. The second one is an eigenvalue equation, written commonly as Hψ = E ψ. Actually there are two different equations known as Schrödinger equation: The first is sometimes called the time-dependent, while the second is called the time independent equation, though in reality, it is simply an equation derived from the first using a mathematical technique known as separation of variables. Austrian physicist Erwin Schrödinger first proposed the equation in early 1926. The Schrödinger equation is one of the fundamental equations of quantum mechanics and describes the spatial and temporal behavior of quantum-mechanical systems.
